Surface tensions
Cochlear implants scaled. ECoG changed the design space. Post 2 of 14 in How BCI got here (2003-2010, The foundations).
By the late 2000s, cochlear implants had become common. Hundreds of thousands of people worldwide had them. They were FDA-approved, covered by insurance, and standardized in surgery. Competing companies manufactured them. Meanwhile, another electrode approach was quietly gathering data that would change BCI design: grids of electrodes on the brain’s surface, recording unexpected signals.
In this post, I’ll discuss two trends from 2007 to 2010 that seemed unrelated then. The cochlear implant story shows how a neural interface matures. The ECoG story reveals a scientific surprise: the cortical surface carries richer signals than scalp EEG. This finding opened new design possibilities that the field still explores.
Detailed source material is available on the bci0 research spine, particularly the 2007–2010 yearly reviews.
The cochlear implant precedent
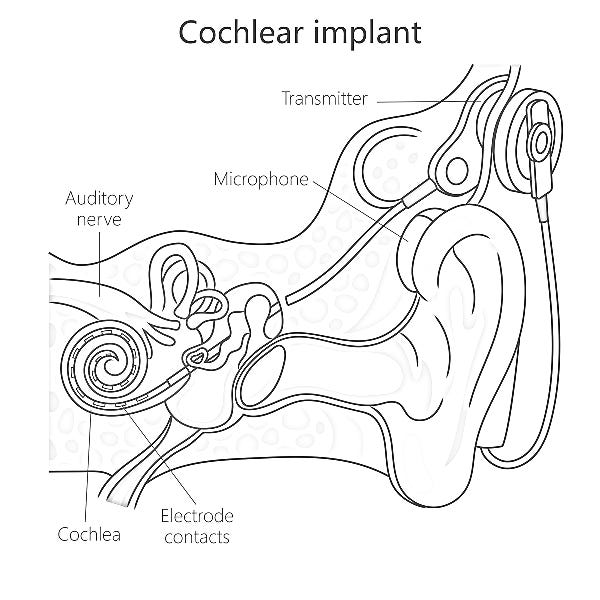
Cochlear implants aren’t as flashy as intracortical BCIs. By the 1990s, the technology was mostly established as a microphone in the ear, an external speech processor, and an implanted electrode array stimulating the cochlea. The science was clear. By 2009, over 150,000 people globally had cochlear implants. The Nucleus 5, launched by Cochlear in September 2009, was the smallest behind-the-ear processor at that time. It was backward compatible for existing recipients. Several manufacturers (Cochlear, Advanced Bionics, MED-EL) were regularly updating their products. Insurance covered these devices, and surgeons had standardized protocols. Long-term safety data reached decades.
This story was largely ignored by the BCI field, but it should have been noted.
Cochlear implants worked not just because of the electrode or stimulation algorithm. They relied on a complete clinical framework: a clear patient population, measurable outcomes (like speech intelligibility), standardized surgery, and reimbursement options through Medicare and private insurers. The commitment to backward compatibility with the Nucleus 5 allowed implant recipients to upgrade without surgery. This was a significant engineering decision that signaled implants were a long-term investment.
The gap between cochlear implants and cortical BCIs is clear: cochlear technology interfaces with the compact, organized auditory nerve. In contrast, the motor cortex is far more complex. Still, the cochlear implant example proved that a long-term neural interface could be safe, manufacturable at scale, and economically viable. By 2010, DBS had reached about 80,000 patients worldwide for conditions like Parkinson’s disease and essential tremor. Together, these devices were the closest thing the neural interface field had to a clinical roadmap.
The core challenge for the BCI field during this time (and at present) was that no motor BCI had met these critical thresholds. There was no reimbursement pathway, no FDA approval for chronic cortical devices intended for home use, and no commercial company with sustainable revenue. The cochlear implant success highlighted this gap.
ECoG and the unexpected signal
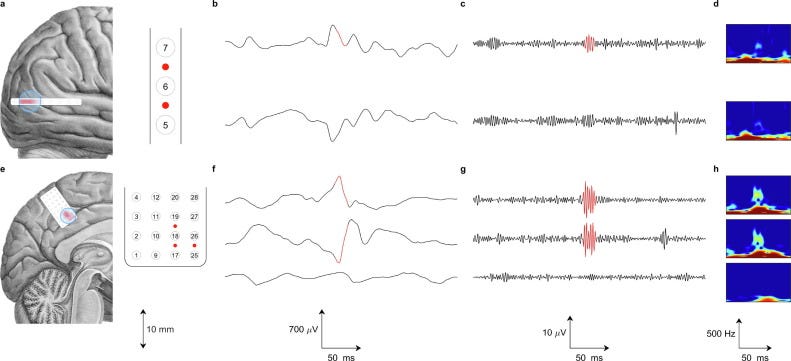
The first clear evidence that electrocorticography (ECoG) could do more than previously assumed came in June 2004. Eric Leuthardt and his team at Washington University in St. Louis published in the Journal of Neural Engineering that four epilepsy patients could control a one-dimensional cursor using gamma-band ECoG signals. Training took three to twenty-four minutes, with success rates between 74% and 100%.
This result was striking for two reasons. First, the training time was much shorter than EEG-based motor imagery BCIs, which required weeks of practice. The ECoG method worked in minutes, indicating that the signals were cleaner. Electrodes placed on the surface of the brain provided higher signal-to-noise ratio and less interference than electrodes on the scalp. Second, ECoG captured high-gamma oscillations at 60–200 Hz, frequencies that carried substantial information about cortical processing, which conventional EEG recordings missed.
By 2005 and 2006, other researchers confirmed these findings. Gerwin Schalk and colleagues at the University of Washington extended the study to ten patients, showing that all subjects achieved control with accuracies between 73% and 100%. Their results revealed that ECoG control worked not just with motor imagery, but also with sensory imagery, speech imagery, and auditory cortex activation. This meant that even patients with extensive motor cortex damage might find usable signals on the cortical surface.
The comparison with intracortical recording is important. The Utah Array’s 96 electrodes penetrate 1–1.5 mm into the cortex, recording from individual neurons. This offers extraordinary resolution, allowing for real-time tracking of the activity of multiple neurons. However, penetration triggers an inflammatory response: microglia activate, leading to scar tissue formation and signal degradation. The Nagle trial had shown this degradation within months.
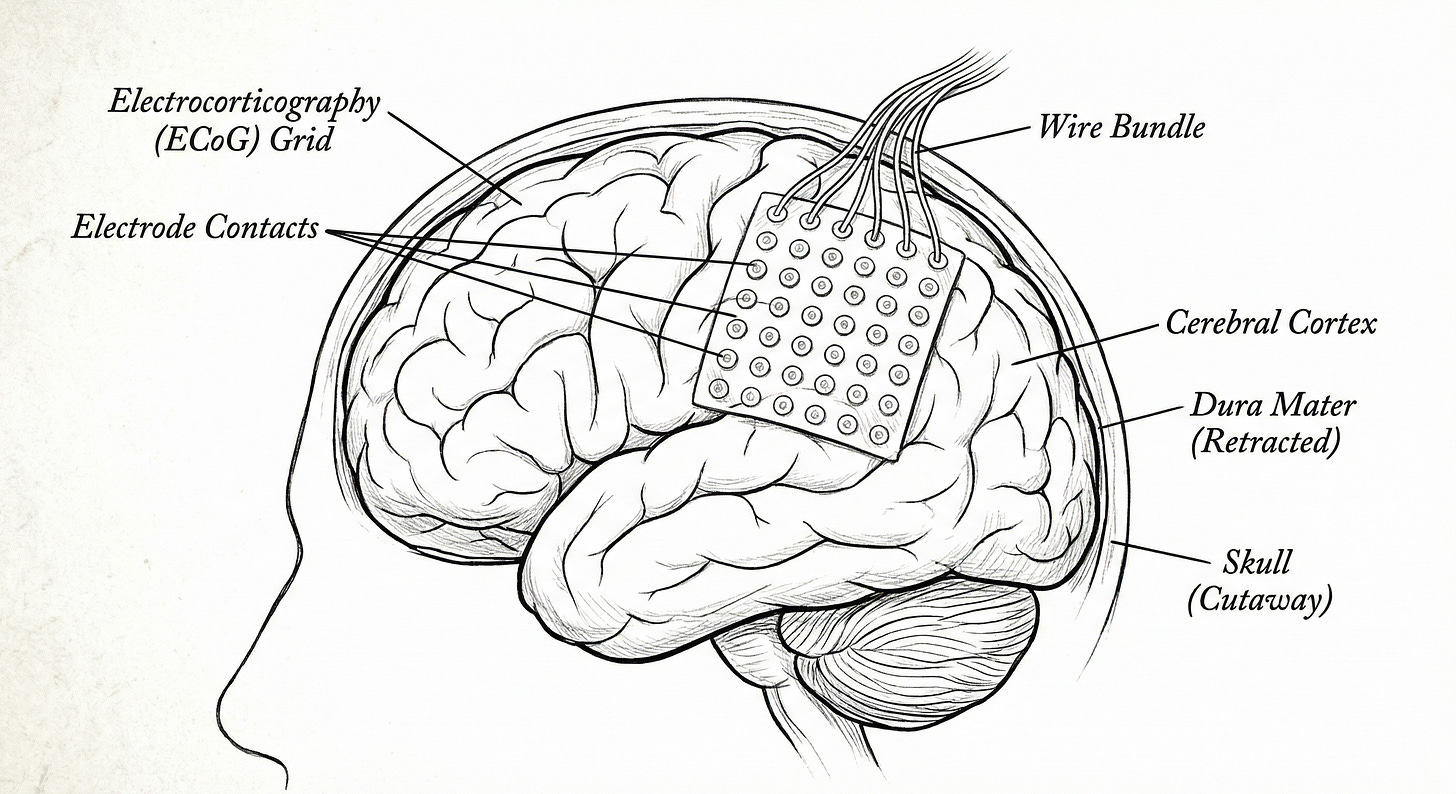
ECoG electrodes sit on the cortical surface without penetrating it. They record summed field potentials from thousands of neurons below each electrode. While spatial resolution is coarser, the inflammatory response is much less severe, showing more chronic stability. The cortical surface did not scar the electrode like the tissue interior did.
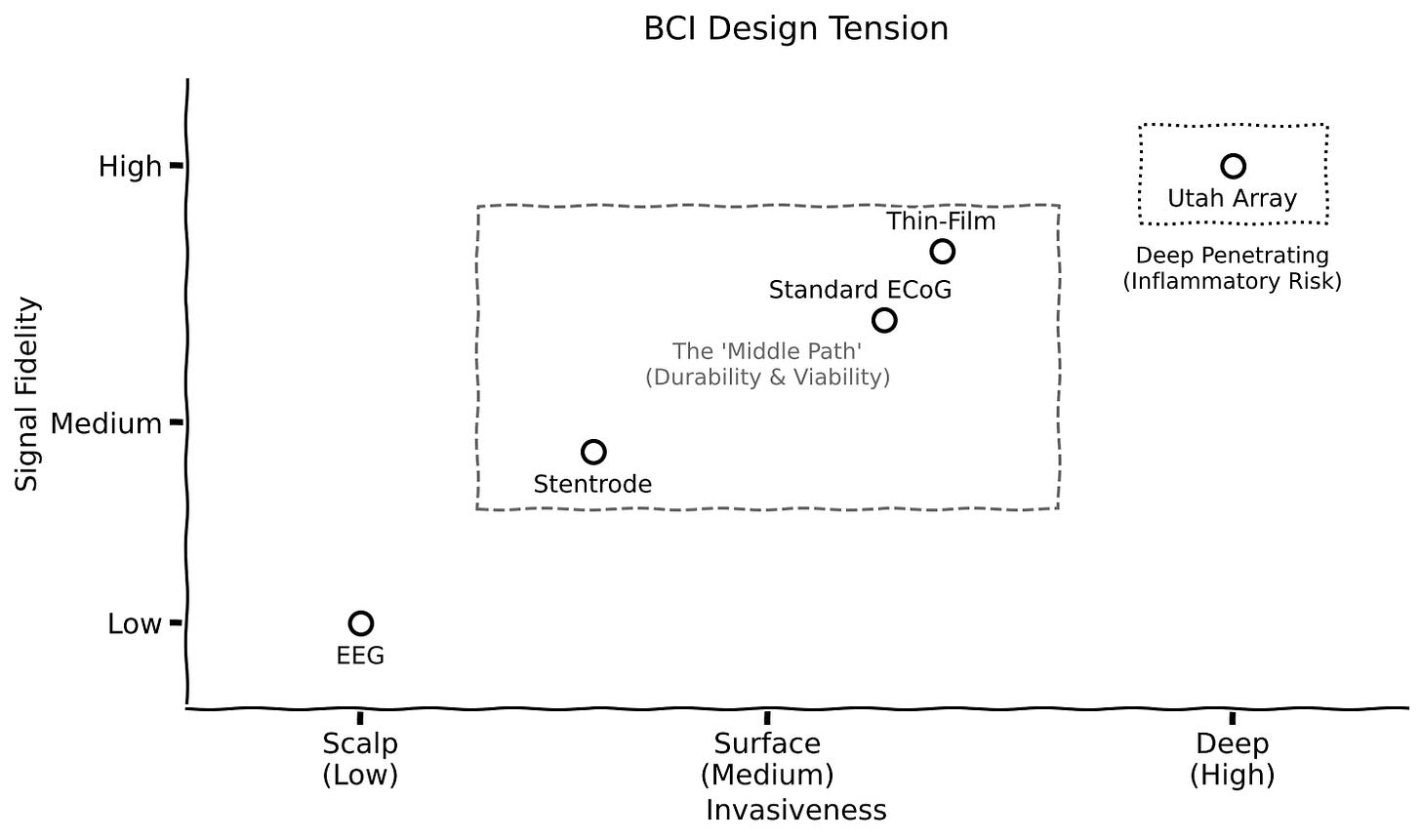
By 2009, Schalk and his team had demonstrated two-dimensional ECoG cursor control with accuracy comparable to single-unit intracortical systems. ECoG could decode two independent control dimensions with performance approaching that of the Utah Array, but without penetrating the cortex. This result clarified the “middle path” argument: ECoG offered better signal than scalp EEG and less surgical risk than intracortical arrays… but still required surgery.
Why the combination mattered
These two trends:
cochlear implants at scale
ECoG as a middle-path modality
were important for the field’s self-understanding.
Cochlear implants illustrated that the design requirements for a neural interface were well-known. A viable device must work for patients at home, not just in labs. Surgical procedures must be standardizable across many centers. Reimbursement must make the economics feasible for hospitals and patients. The hardware must last decades, and software should be updatable without surgery. None of these were research challenges; they were engineering, regulatory, and business issues. Yet by 2010, no motor BCI had addressed any of these.
ECoG revealed that the design possibilities for cortical BCIs were broader than the intracortical-versus-EEG binary suggested. A middle path offered better signal fidelity than scalp recordings without the chronic issues that made Utah Array longevity uncertain. The results from Leuthardt and Schalk began pointing toward speech and language decoding, where the cortical surface has dense, organized functional representations that ECoG could utilize. This connection would become a productive thread in BCI research for the next decade.
The key insight was the design tension the field began to articulate: go deep for higher resolution, accepting inflammatory risks; or stay on the surface for durability, accepting lower resolution. This tension remains unresolved but has been productively explored. The Stentrode from Synchron addresses this; it is an electrode array mounted on a stent deployed into a cerebral blood vessel. Precision Neuroscience’s thin-film subdural array offers another answer. Both products from the 2020s stem from the ECoG insights of 2004–2010: penetration isn’t always necessary for useful signals.
What was still uncertain
While ECoG’s chronic stability looked better than intracortical arrays, it was largely untested over long periods relevant to BCI use. The epilepsy monitoring studies that generated most ECoG BCI data involved temporary implants lasting a few weeks. It remained unknown if a chronic subdural ECoG implant would remain stable over years without gliosis, electrode-tissue degradation, or clinical complications. No approved chronic ECoG devices existed… yet.